The new super meta review of Ivermectin is out. It’s 27 pages of fine print detail and 144 references, and it’s very impressive.
Bryant et al soaked themselves in 24 studies involving 3,406 people and found that ivermectin use reduced deaths by a very nice 60% with “moderate certainty”. But ivermectin appears to be at its best when used to prevent infections in the first place. There was “low certainty” but with prophylactic use Covid infections were reduced by an average of 86% . But by the time patients were “in need of mechanical ventilation”, the data, while muddy, suggested ivermectin was not much help.
The bad thing about Ivermectin is that there are not many bad things. It’s too good, too cheap, too safe, and too far out of patent to be profitable.
Given the evidence of efficacy, safety, low cost, and current death rates, ivermectin is likely to have an
impact on health and economic outcomes of the pandemic across many countries. Ivermectin is not a new
and experimental drug with an unknown safety profile. It is a WHO “Essential Medicine” already used in several different indications, in colossal cumulative volumes.
We can almost hear the frustration as Bryant et al compare how easily other drugs get approval to treat Covid:
Corticosteroids have become an accepted standard of care in COVID-19, based on a single RCT of dexamethasone.1 If a single RCT is sufficient for the adoption of dexamethasone, then a fortiori the evidence of 2 dozen RCTs supports the adoption of ivermectin.
RCT means a Random Controlled Trial. Sometimes one trial will do, other times 24 trials isn’t enough.
h/t to Ian and Phillip
Meanwhile, the BBC has announced that Ivermectin will be studied as possible treatment in UK
Hold off on the champers. The Principle Study appears to be a holding pattern to fend off questions about “why they weren’t studying Ivermectin”. Good reporters will want to ask why the trial will allow people to join up to 15 days after they get symptoms when the best results come with early treatment and even prophylactic use. If only there were good reporters left at the BBC.
The trial will also allow people who are vaccinated to join which is a good way to dilute the results. If we compare 2000 vaccinated people with 2000 also-vaccinated people the difference between placebo and drug may not be very significant. And if the vaccine “is the ticket to freedom” why invite vaccinees to join the trial? Is it, perhaps a back route to reducing deaths in vaccinees who get breakthrough infections? Let’s just hope for the vaccinees sake, that the docs don’t wait until they’re on the ventilator.
If the UK Ministry of Big Pharma Health really wanted to test Ivermectin, they’d offer it to people in high risk areas who weren’t sick and weren’t vaccinated, and compare like for like cases without Ivermectin in the same area.
But that might show good results.
h/t Steve,
Indonesia however is poor enough to approve Ivermectin use. Without access to $5000-dollar-a-day ICU beds, or mass experimental vaccines, Indonesia is looking down the Delta variant barrel. They have been given Sinovax, the Chinese vaccine, but seemingly that’s not much (as it wasn’t it the Seychelles, or in Chile either).
Production capacity of Ivermectin is said to be 4 million per month. (Presumably they refer to doses?). But with 270 million people, that might not be enough, even if they are dished out to the highest risk people. With 15,000 new daily cases and doubling fast, they are racing against the tide. They would probably need a few million doses just for the current contacts.
Indonesia doesn’t just have two new variants of concern, they have 211.
Watch this space…. (and pray for the people of Indonesia). We have lift off. 🙁
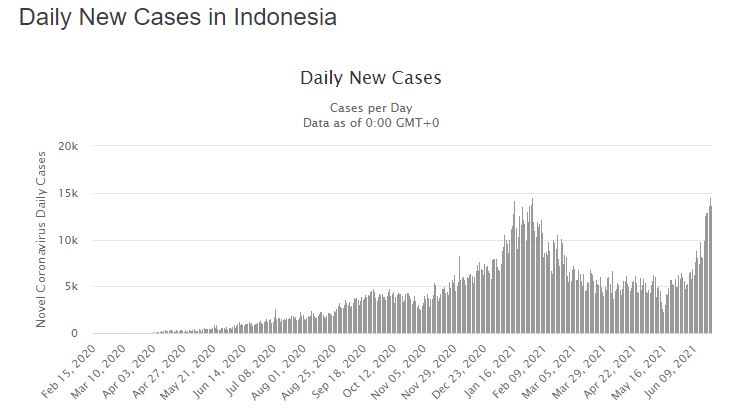
Things are rapidly getting out of hand in Indonesia.
We have to admire both Craig Kelly and Malcolm Roberts in Australia.
REFERENCES
Bryant, Andrew MSc1,*; Lawrie, Theresa A. MBBCh, PhD2; Dowswell, Therese PhD2; Fordham, Edmund J. PhD2; Mitchell, Scott MBChB, MRCS3; Hill, Sarah R. PhD1; Tham, Tony C. MD, FRCP4 (2021) Ivermectin for Prevention and Treatment of COVID-19 Infection, A Systematic Review, Meta-analysis, and Trial Sequential Analysis to Inform Clinical Guidelines
American Journal of Therapeutics: June 17, 2021 – Volume Publish Ahead of Print – Issue –
doi: 10.1097/MJT.0000000000001402